
The key findings are that the para-doped Mn catalyst affords excellent NH 3 selectivity and NRR activity with an extremely low free energy barrier of 0.36 eV. If its atomic weight is 70.4 g/mol and the atomic radius is 0.126 nm, compute its density. A hypothetical metal has the simple cubic crystal structure shown to the right. Finally, using these two catalysts, the energetics of the whole NRR pathway were calculated. Question: Calculate the radius of an iridium atom, given that ire has an FCC crystal structure, a density of 22.4 g/cm3, and an atomic weight of 192.2 g/mol. Mn and Fe were determined to have high catalytic activities for the NRR based on the screening of the adsorption energies and activation of N 2, formation of adsorbed N 2H, desorption of adsorbed NH 3, and HER inhibition. In this study, density functional theory calculations were used to investigate the performance of NRR electrocatalysts based on the transition metal doped Ir(100) surface to yield two-atom active sites (TM from an energetic perspective. To enable the use of the NRR in an industrial setting, high-performance electrocatalysts are required. Therefore, current implementations of the NRR are not suitable for industrialization. However, the efficiency of the NRR is limited by the high N N bond strength and the competitive hydrogen evolution reaction (HER). It has an electron configuration of Xe6s 2 5d 7. The element iridium is the twenty-sixth element in the transition metals. 
The elements rhodium and meitnerium are above and below iridium. It is located in the d-block of the periodic table, between osmium and platinum. The electrocatalytic nitrogen reduction reaction (NRR) is an example of such a reaction. Iridium has the atomic symbol Ir with an atomic number of 77. Because of the growing demand for NH 3 in industry and agriculture, the development of alternative technologies, especially those having low energy consumption and producing little pollution, is urgently required. 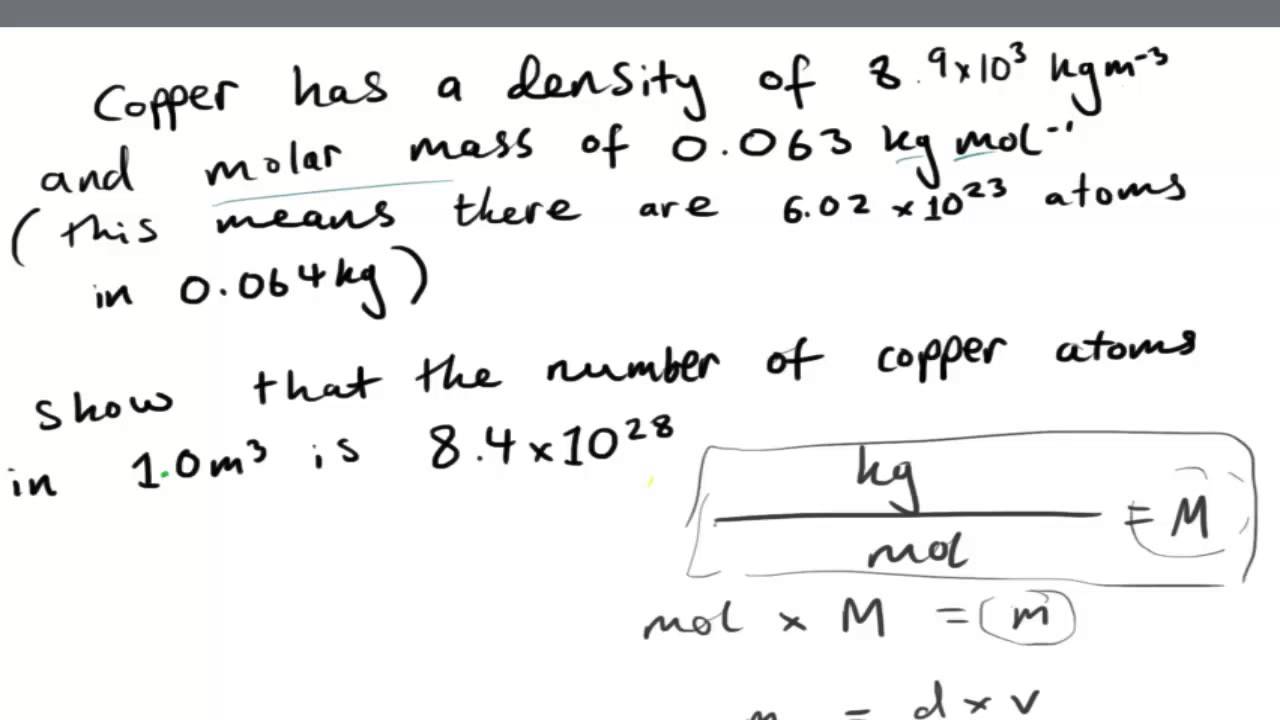
The main industrial process for the production of NH 3 is the Haber–Bosch process, which requires a large amount of energy as a result of the use of high temperatures and pressures, making it costly, inefficient, and environmentally unfriendly. Ammonia (NH 3) is one of the most important basic chemicals and is used worldwide in both industry and agriculture.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
